Natural and artificial sweeteners have become increasingly common in modern diets, often replacing sugar for health, metabolic, or culinary reasons. While both types aim to provide sweetness with fewer calories, they differ significantly in their origin, chemical structure, metabolic processing, and effect on blood glucose. Understanding these distinctions is essential for informed dietary decisions, especially for those managing weight, diabetes, or metabolic conditions.
Chemical Composition: Natural vs Synthetic Origins
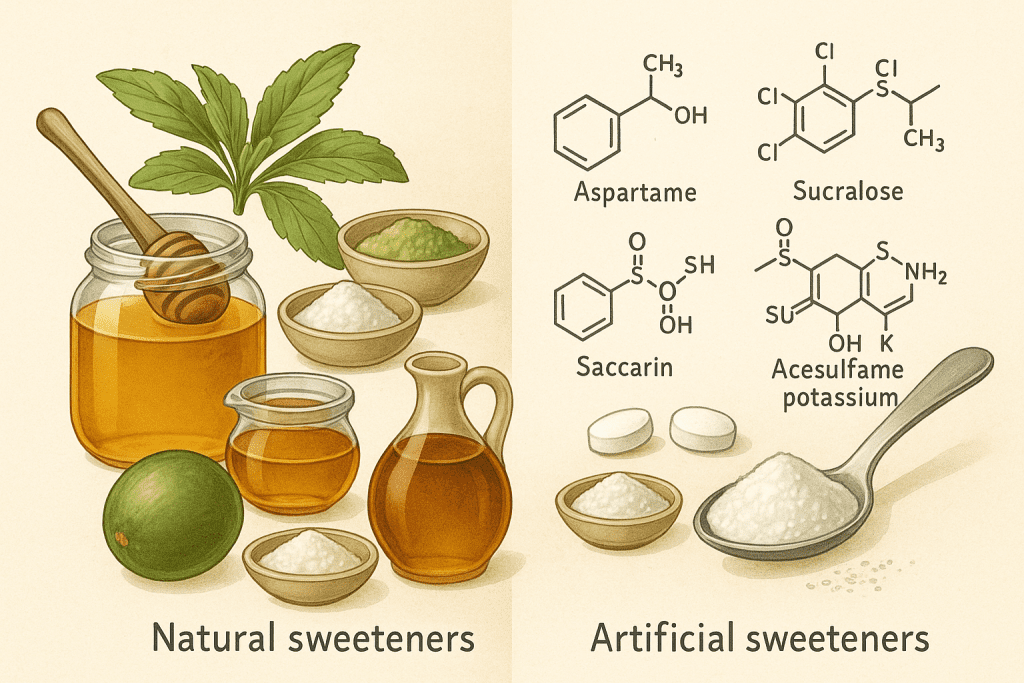
Natural sweeteners are substances derived directly from plants or natural sources with minimal processing. These include honey, maple syrup, agave nectar, and purified compounds like stevia (extracted from Stevia rebaudiana) and monk fruit (from Siraitia grosvenorii). Chemically, many of these sweeteners contain mono- and disaccharides or glycoside compounds like stevioside and rebaudioside A in stevia.
Artificial sweeteners, on the other hand, are synthetic compounds engineered to mimic the sweetness of sugar, often with hundreds of times the intensity. Common artificial sweeteners include:
- Aspartame (a methyl ester of a dipeptide)
- Sucralose (chlorinated derivative of sucrose)
- Saccharin (a sulfonamide compound)
- Acesulfame potassium (a potassium salt of a synthetic organic acid)
These compounds are not structurally similar to sugars but stimulate the same taste receptors on the tongue. Because of their potency, only trace amounts are needed, which is why most artificial sweeteners contribute negligible calories.
Metabolism and Absorption
The human body metabolizes each sweetener differently based on its structure.
Natural Sweeteners:
- Honey, agave, and maple syrup are metabolized similarly to glucose and fructose — absorbed in the small intestine, processed in the liver, and provide energy (typically 3–4 kcal/g).
- Stevia compounds (e.g., rebaudioside A) are not digested in the small intestine. They pass to the colon, where gut microbiota degrade them into steviol, which is absorbed and excreted via urine. They contribute virtually no energy.
Artificial Sweeteners:
- Aspartame is metabolized into its amino acid components (phenylalanine, aspartic acid, and methanol) and absorbed. Despite its caloric content (4 kcal/g), the amount used is so small that the total energy contribution is minimal.
- Sucralose is largely excreted unchanged in feces, with only a small amount absorbed and excreted via urine. It is poorly metabolized by humans.
- Saccharin and acesulfame K are not metabolized at all — they are absorbed and excreted unchanged in urine.
This variation affects not only energy intake but also the compounds’ interaction with the gut microbiome and long-term metabolic outcomes.
Glycemic Impact and Insulin Response
One of the most critical distinctions between sweeteners is their glycemic index (GI), a measure of how rapidly a substance raises blood glucose levels.
Table: Glycemic Index & Sweetness Relative to Sucrose
| Sweetener | Type | GI (approx.) | Sweetness vs Sucrose | Calories (per gram) |
|---|---|---|---|---|
| Glucose | Natural sugar | 100 | 0.7 | 4 |
| Sucrose | Natural sugar | 65 | 1.0 | 4 |
| Fructose | Natural sugar | 15 | 1.3 | 4 |
| Honey | Natural | 50–70 | 1.0–1.2 | ~3 |
| Agave syrup | Natural | 10–20 | 1.4 | ~3 |
| Stevia (extract) | Natural | 0 | 200–300 | 0 |
| Aspartame | Artificial | 0 | 180–200 | 4 (negligible) |
| Sucralose | Artificial | 0 | 600 | 0 |
| Saccharin | Artificial | 0 | 300–500 | 0 |
| Acesulfame K | Artificial | 0 | 200 | 0 |
Most artificial sweeteners have a GI of zero, meaning they do not trigger any significant rise in blood glucose or insulin response. Natural sweeteners vary: fructose has a low GI but is metabolized differently than glucose, while honey and maple syrup cause moderate spikes.
However, some studies suggest a paradoxical metabolic effect of certain artificial sweeteners. For example, sucralose and saccharin may influence insulin sensitivity or alter gut microbiota, indirectly affecting glucose regulation. These effects are still under investigation and vary by individual and duration of consumption.
Safety, Toxicology & Regulatory Status
Both categories of sweeteners are widely consumed and regulated by food safety agencies like the FDA (U.S.), EFSA (EU), and WHO.
Natural Sweeteners:
Most natural sweeteners, such as stevia, have GRAS (Generally Recognized as Safe) status in the U.S. However, raw or unprocessed extracts may contain bioactive compounds with unpredictable pharmacological effects. For instance, crude stevia extracts were previously banned due to insufficient toxicological data.
Artificial Sweeteners:
Artificial sweeteners have undergone decades of testing. Key findings:
- Aspartame is safe for the general population but must be avoided by individuals with phenylketonuria (PKU) due to its phenylalanine content.
- Sucralose is heat-stable but may degrade at very high temperatures (>120°C), potentially forming chlorinated byproducts in cooking.
- Saccharin was once linked to bladder cancer in rodents, but subsequent studies showed no clear risk in humans at typical consumption levels.
Each sweetener has an established ADI (Acceptable Daily Intake), measured in mg/kg body weight/day. Staying within these limits ensures long-term safety.
Use Cases in Diet, Cooking & Industry
Choosing a sweetener often depends not just on health impact but also on functionality in recipes and commercial food production.
Heat Stability & Solubility
Some sweeteners break down or lose sweetness when heated. For example:
- Aspartame degrades in heat — not suitable for baking.
- Sucralose, stevia, and acesulfame K are heat-stable and commonly used in baked goods and soft drinks.
Taste Profile
Natural sweeteners like stevia can leave a bitter or licorice-like aftertaste, especially at high concentrations. Artificial sweeteners vary:
- Saccharin has a metallic aftertaste.
- Sucralose has a more sugar-like flavor, making it popular in beverages.
Culinary Substitutions: Because artificial sweeteners are so potent, they are often blended with bulking agents (like maltodextrin) to mimic sugar’s volume and mouthfeel.
Sample use guide for baking:
- For sugar replacement in cakes: Use sucralose blends for 1:1 substitution.
- For low-carb desserts: Try stevia + erythritol mixtures to balance taste and structure.
- For sauces or drinks: Liquid stevia, monk fruit, or acesulfame K offer good solubility.
Industry Applications
- Soft drinks: Aspartame, sucralose, acesulfame K.
- Protein bars and shakes: Stevia, sucralose.
- Baked goods: Sucralose blends, polyol combinations (erythritol, xylitol).
In recent years, there has been a trend toward blended sweetener systems, combining natural and synthetic components to optimize taste, texture, and calorie control.
Conclusion
Natural and artificial sweeteners serve overlapping but distinct roles in modern nutrition. Natural options offer a closer link to whole foods and simpler metabolism, but often come with calories and variable glycemic effects. Artificial sweeteners, while chemically different and extremely potent, allow for calorie-free sweetness with no direct glycemic load — though their long-term metabolic effects remain under study.
A balanced approach lies in contextual use: understanding each sweetener’s properties and applying them where appropriate. This requires considering not only calories and GI but also cooking application, taste profile, digestive tolerance, and individual health goals.
In the evolving landscape of dietary science, sweeteners — both natural and artificial — represent tools, not solutions. Used wisely, they can support healthier lifestyles without sacrificing the sensory pleasures of sweetness.